
Vitamin E is a fat-soluble compound along with A, D, and K. Said vitamin is most widely recognized by its anti-oxidant capabilities and capacity to maintain and protect cell membranes and lipoproteins (Gropper, Smith, & Carr, 2018). As a means of appreciating vitamin E’s role as an anti-oxidant, the following will briefly consider its structure, function, and protective relationship with red blood cells (RBCs) and mitochondria.
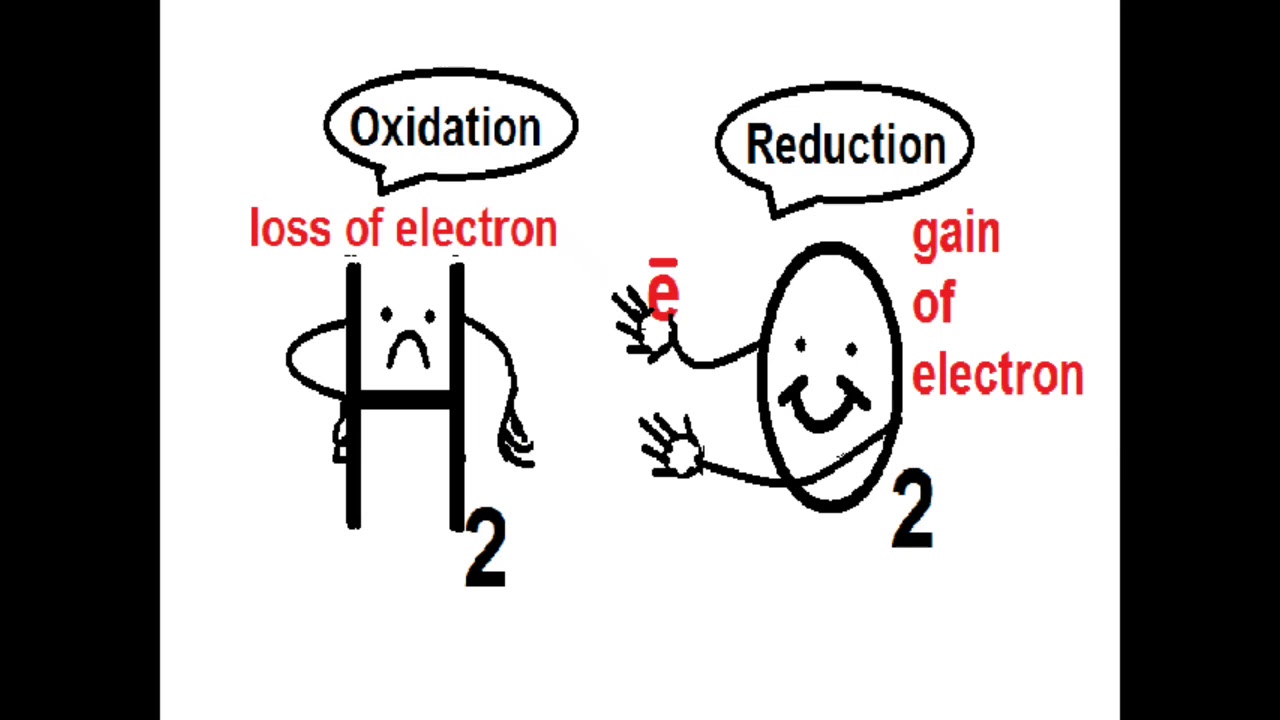
Vitamin E maintains cell membrane integrity by way of preventing the oxidation (i.e., loss of electrons) of unsaturated (at least one double bond in the chain) fatty acids within the membrane wall (Gropper et al., 2018). Tissues and organs that are especially susceptible to oxidation include the brain, erythrocytes (RBCs), and lungs (Gropper et al., 2018). RBC’s, for example, are particularly vulnerable to oxidation due to their close proximity to oxygen molecules and because of their lipid architecture; RBCs are high in polyunsaturated fatty acids, increasing their susceptibility to oxidation (Gropper et al., 2018). The following will consider oxidative compounds and their interactions with vitamin E in the body in greater detail.
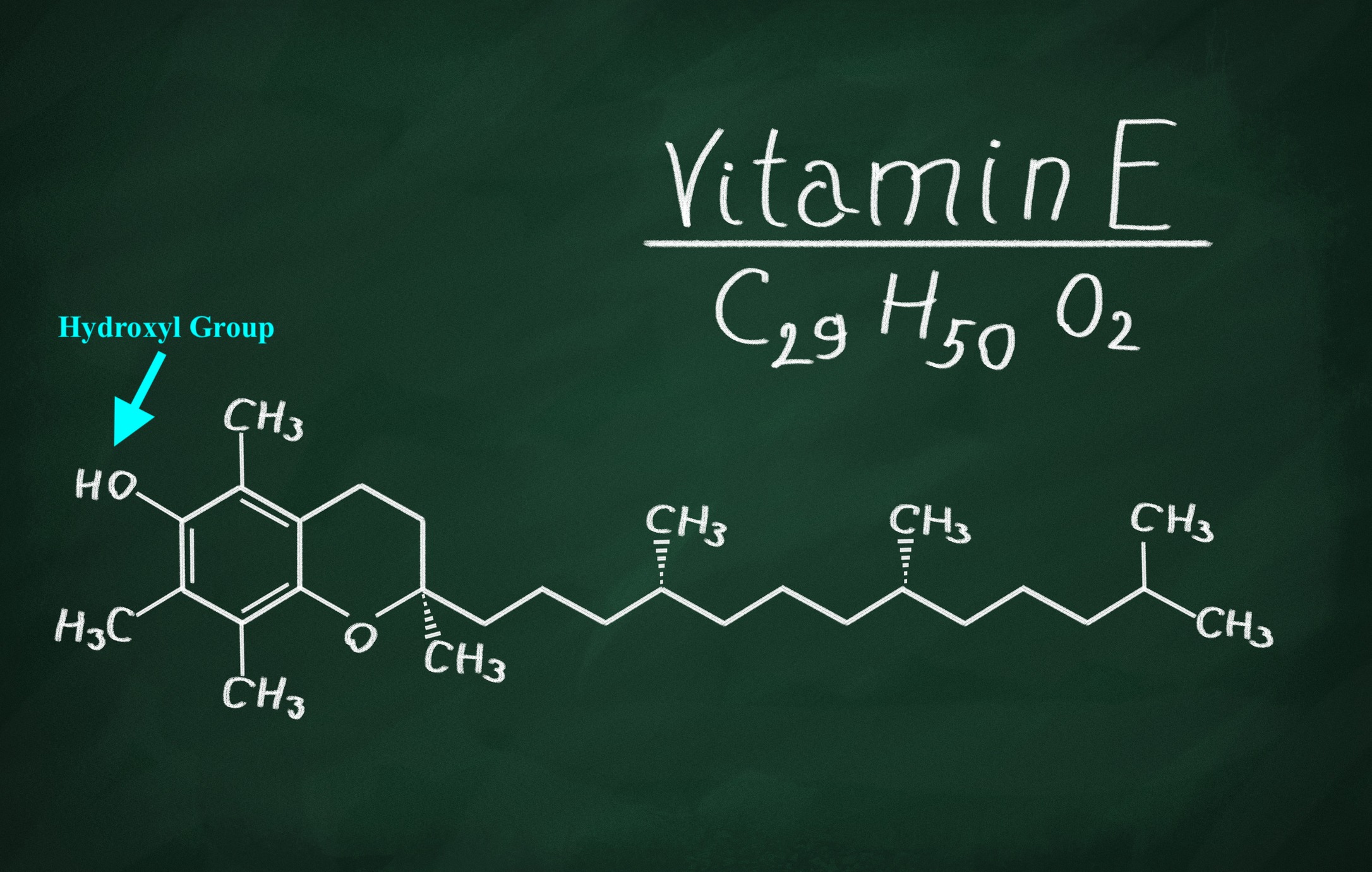
Singlet molecular oxygen (SMO), as described by Gropper et al. (2018), is a destructive reactive oxygen species (ROS) molecule than has an affinity for oxidizing protein, lipids, and DNA. Said molecule is formed within the body from multiple processes to include photochemical reactions, lipid peroxidation of membranes, and enzymatic reactions (Gropper et al., 2018). In essence, SMO “takes” electrons from other substances, and such reactions chemically disrupt the structure and function of the targeted substance. Vitamin E can help inhibit such oxidative reactions that ROS (to include superoxide anion radical, singlet oxygen, hydroxyl radical, and perhydroxyl radical) inflicts (Gropper et al., 2018). Vitamin E contains within its structure, a hydroxyl group that has the capacity to donate hydrogen ions to an ROS and reduce/inhibit the oxidation of fragile cell membrane structures (Gropper et al., 2018). Once enough hydroxyl groups are presented to ROS, cell membrane destruction decreases; a process is known as termination (Gropper et al., 2018).
Termination is key in protecting membranes as uncontrolled oxidation can cause hemolysis (cell membrane destruction) in RBCs (Gropper et al., 2018). Vitamin E also plays an intricate role with mitochondria; a well-known source, during oxidative metabolism, of free radical production (i.e., superoxide free radicals) (Beal, 1996). Left unchecked, ROS can build and disturb functioning of the mitochondria leading to conditions such as Alzheimer’s disease, Huntington’s disease, Parkinson’s disease, diabetes, and fatty liver disease (Mao, Kraus, Kim, Spurlock, Bailey, & Beitz, 2011). Vitamin E can assist in controlling ROS damage from said organelle by way of electron donation (Mao et al., 2011).
As evidenced in previous sections, ROS can have destructive outcomes if not tightly regulated within the human body. Vitamin E is a fat-soluble molecule that, in conjunction with other micronutrients/macronutrients, can assist in blunting the effects of ROS. Ultimately, such inclusion of said micronutrient can be part of optimizing individuals’ overall health, performance and longevity.
References
Beal, M. F. (1996). Mitochondria, free radicals, and neurodegeneration. Current Opinion in Neurobiology, 6(5), 661-666.
Gropper, S. S., Smith, J. L., & Carr, T. P. (2018). Advanced nutrition and human metabolism (7thed.). Boston, MA: Cengage Learning.
Mao, G., Kraus, G. A., Kim, I., Spurlock, M. E., Bailey, T. B., & Beitz, D. C. (2011). Effect of a mitochondria-targeted vitamin E derivative on mitochondrial alteration and systemic oxidative stress in mice. British Journal of Nutrition, 106(1), 87-95.
-Michael McIsaac
